Defect of the Month
Back to AGR's LibraryPerhaps nothing says “cozy” like an image of grandma in a rocking chair, knitting a blanket with a big basket of yarn at her feet. The bunches of crystals in this Scanning Electron Microscope image appear remarkably similar to jumbled skeins of yarn, but have a very different source. They were found inside a molybdenum blister created in the glass melt when boosting electrodes were exposed to air in the furnace. The crystals themselves are composed primarily of sodium molybdate with a smaller amount of calcium molybdate.
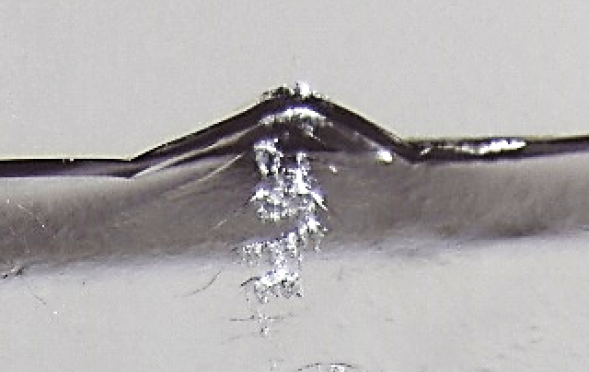
While this might appear to be a road to a mountain, it is a magnified view of the horizontal crack revealed that the origin was related to a vertical scuff mark with a few associated crescent cracks. Further investigation led to the conclusion that the scuff marks had been created by the downward sliding contact of the heel of one bottle against the fractured bottle. This action most likely had occurred during case packing.

Have you ever wondered what was in the dust sucked up by your vacuum cleaner? At a glass plant, the profusion of compressed air lines, exhaust ducts, and cooling air means that there is a lot of air in circulation, all of which can contribute to a dusty environment. Filters are used to minimize the dustiness, and those filters sometimes become clogged. This EDX composition map shows the contents of one such filter. In an optical microscope, the dust looks like dirty sand, but the spectral map easily discerns between glass fragments, limestone, and soda ash. All three are batch materials used for making glass. The small amount of chlorine is due to salt.

Here in the United States, the fast food chain Chick-fil-A is renowned for their delectable fried chicken offerings. With just the right amount of crispy breading, their chicken nuggets are perfect for dipping into a variety of sauces, including the author’s favorite –Polynesian. This image of a stone in glass resembles a (rather bleached) nugget at the precise moment before being dunked into a (rather colorful) sauce. The “crispy breading” is comprised of small nodules of zirconia (ZrO2), which were created by the interaction of the glass melt with an AZS refractory. The glassy phase of the refractory dissolves over time, creating an alumina-rich solution sac and a stone of nodular zirconia. The “sauce” in this image is the induced stress in the surrounding glass, rendered visible with polarized light.
Pagination
- Page 1
- Next page